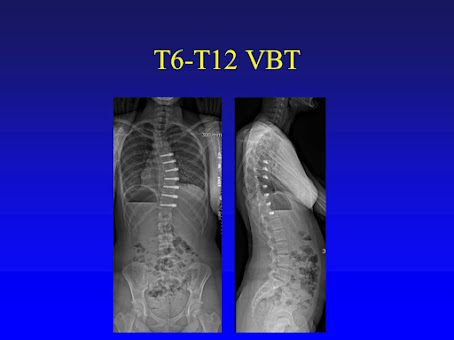
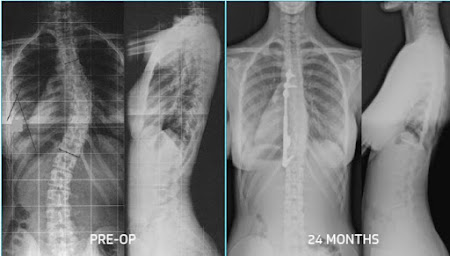
In previous posts the Anterior Vertebral Body Tethering (VBT) has been presented. This implant utilizes the flexibility of the growing thoracic spine, and its growth, to straighten the spine and then modulates its growth through the remainder of spinal growth. There has been a lot of social media interest and publications about this procedure over the last several years touting its benefits, which are small incisions, rapid recovery, and the ability to correct
the spine deformity yet not fuse the spine which preserves some of the spinal
motion. The challenges of this procedure have also been well-documented, such
as how much tension to apply to the tether/spine, how many levels to tether,
and chronologic timing of the procedure (when to do the procedure).
Interestingly, an alternative
procedure was approved the same month (8/2019), the ApiFix device. There has been a lot less attention give to
the ApiFix device, compared to the VBT procedure. So the question is why?
Why has all the focus been on
VBT and not ApiFix?
Some reasons:
1. 1. VBT has been used
by pediatric spine surgeons in the U.S. since the 2010s, although in an
unlabeled manner. Whereas ApiFix was
developed in Israel and used only outside the U.S. Hence more surgeon in the U.S. are familiar
to the VBT procedure than ApiFix
2. 2. To many the
ApiFix looks and acts similar to a traditional growing rod. Whereas the VBT is inserted through small incisions,
placed on the anterior spine and uses a tether as opposed to a lot of metal.
3. 3. Due to the longer
surgeon experience with VBT, dramatically more research has been produced on
VBT.
4. 4. Currently, any
pediatric spine surgeon who undergoes a short training session can perform the
VBT procedure. For the ApiFix device the
company is performing a study on all patients in the U.S., so all surgeons who
use the device undergo more rigorous training and all patients will be
studied. This means there are far fewer
surgeons in the U.S. performing the ApiFix surgery than VBT
5. 5. As a continuation
of #4, since all patients are going to be in the FDA study on ApiFix there is
strict criteria as to who can undergo the procedure. The VBT procedure is left up to each surgeon
to determine is applicability and effectiveness for each patient.
The effectiveness of VBT and
ApiFix for each patient requires an in-depth discussion between patient/family
and the surgeon. At present there is no
scientific evidence of the superiority of either system